Mind the Gap!

BCI technology is racing from research labs into clinical and everyday applications never before seen.
On a spring morning in Beijing, Zhang Xin sat in his office, busy preparing for his next popular science livestream centered on brain function monitoring. As chairman of CASIBRAIN Technology, this researcher-turned-entrepreneur working at the intersection of brain science and medical devices embodies the pragmatic spirit of China’s scientific community: precise, solution-oriented and skilled at translating the most futuristic technologies into practical clinical tools.
“Brain-computer interface (BCI) sounds impressive, but for ordinary people, it has to solve real problems,” Zhang told Beijing Review in an exclusive interview.
BCI establishes a direct information pathway between the brain and an external device, enabling two-way communication by decoding neural signals. The global industry generally categorizes BCI into three technological types: non-invasive, semi-invasive and fully invasive, each with substantial differences in safety, signal precision and clinical application scenarios.
Born out of the Chinese Academy of Sciences’ (CAS) Institute of Automation—China’s premier national scientific research institution—CASIBRAIN Technology has built a core technology system integrating BCI and AI, drawing on over two decades of CAS brain research. The company is dedicated to translating brain science into clinical practice.
Its non-invasive, whole-brain near-infrared functional brain monitoring system has already received an innovative medical device designation from China’s National Medical Products Administration (NMPA) and is now in full clinical use, benefiting a growing number of patients.
In today’s BCI landscape, grand visions of “mind-to-mind communication” and “human-machine symbiosis” are abundant. Yet Chinese teams remain firmly grounded, focusing on clinical pain points.
The year 2026 marks a pivotal moment for China’s BCI industry. The past decade was defined by long-term, patient research and development (R&D) inside laboratories; this year, with innovative products reaching the market and new pricing models being rolled out, the technology is racing from research labs into clinical and everyday applications never before seen.
Diagnostic precision
For Zhang, the primary function of a BCI isn’t “control”—it’s “understanding.”
“Take epilepsy. Traditional detection methods can tell you that abnormal electrical discharges are happening somewhere in the brain, but pinpointing the exact region is extremely difficult. Each channel on the electrode picks up signals,” Zhang explained. “It’s like an AI-assisted driving system in an electric vehicle. You need LiDAR (light detection and ranging technology) and continuous video input combined to accurately identify objects. Our technology works the same way: By integrating electrical signals with near-infrared functional brain imaging, we can accurately locate abnormal brain regions.”
This diagnostic precision is already reshaping operating rooms across China. In anesthesiology, some hospitals have begun using Electroencephalography (EEG) or brain function monitoring metrics to help assess sedation depth. “Give too much anesthetic, and you risk brain injury; give too little, and the patient may wake up during surgery,” Zhang noted. “With these metrics, doctors can deliver more precise, individualized anesthesia care.” This system has already begun rolling out to primary and community-level medical institutions.
This model of broad-based distribution is a signature feature of China’s healthcare system: breaking down the barriers that have long confined advanced medical technology to elite first-tier hospitals, and pushing cutting-edge devices and treatment protocols down to county-level and community institutions, making healthcare truly inclusive.
At the same time, Zhang acknowledged, “Initially, the reach of these technologies was relatively limited. It’s not just about device safety and pricing; there are also ethical considerations.” As the technology matures and gains clinical acceptance, he believes it will gradually reach more people.
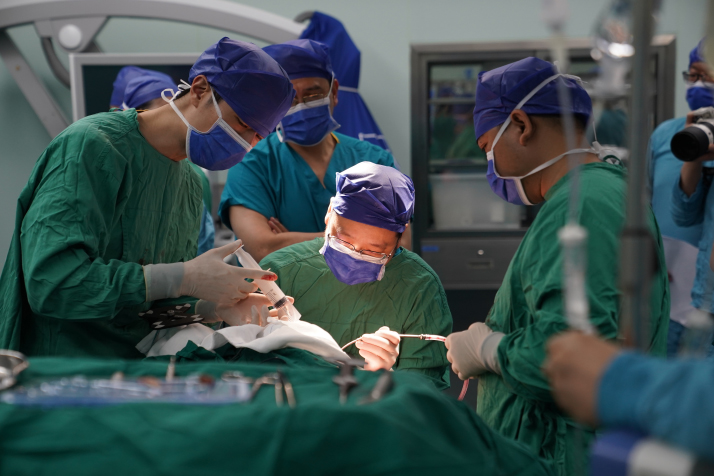
A tale of two philosophies
While Zhang’s CASIBRAIN Technology focuses on non-invasive systems, another Chinese player, Neuracle Medical Technology, the world leader in cost-effective, research-grade EEG systems, among other systems, is grabbing the global spotlight with its implantable BCI. In March, the NMPA approved the registration application for Neuracle’s NEO invasive BCI hand-function rehabilitation system, bringing about the world’s first market launch of an invasive BCI medical device and positioning China at the forefront of clinical BCI application worldwide.
Behind this milestone stands Professor Hong Bo at Tsinghua University, who has spent two decades walking a path few others have taken.
When Hong returned to Tsinghua after academic research overseas, he chose a route that seemed far removed from the mainstream. At the time, the global BCI landscape was dominated by two diverging technological philosophies: One, championed by American billionaire Elon Musk’s Neuralink, pursued a fully invasive approach: direct cortical penetration with electrodes, delivering high neural signal fidelity but also posing significant surgical trauma and risk of immune rejection. The other took a purely non-invasive route, using external electrode caps, which are safe but, degraded by the skull’s natural barrier, lack the resolution and accuracy needed for high-precision clinical applications.
Hong chose the middle path: an epidural approach—placing a thin-film electrode on the dura mater, the tough outermost membrane enveloping the brain and spinal cord, without penetrating or directly contacting brain tissue. This semi-invasive strategy achieves an optimal balance of signal quality and safety.
The approach, tempering ambition with prudence, has already yielded results. In clinical trials involving patients with high-level spinal cord injuries, some have shown substantial improvement in motor function scores after using the NEO system.
Meanwhile, Neuralink and other international players continue to accelerate human trials and seek regulatory approvals overseas, intensifying global competition in BCI R&D and commercialization.
Neuralink has announced plans to mass-produce its invasive BCI devices in 2026, with a fully automated surgical workflow designed to eliminate the need to penetrate the dura mater. On March 25, the company unveiled another breakthrough: its brain chip can now translate neural signals directly into audible speech, enabling people with aphasia or severe speech impairments to “speak” using only their thoughts.
Accessible technology
If scientific breakthroughs are the spark, then 2026’s national policy is the powerful fuel that accelerates China’s BCI engine.
This year’s government work report, approved by the national legislature in early March and which outlines the policy priorities for 2026 and the 15th Five-Year Plan (2026-30) period, for the first time listed BCI as a key future industry for cultivation. Brain science and BCI innovation clusters are emerging across China, from dedicated research centers in Beijing to industrial ecosystems spanning materials, chips, devices and clinical rehabilitation.
But for ordinary people, the most tangible value lies in affordability.
Zhang’s concern that limited popular accessibility would restrict the practical value of emerging medical technologies is being addressed by the National Healthcare Security Administration (NHSA) with remarkable speed. At present, regional authorities are exploring pricing frameworks for BCI-related medical services, laying the groundwork for future coverage under basic medical insurance schemes. China’s basic medical insurance schemes provide coverage for approximately 1.36 billion people, with the coverage rate currently above 95 percent of the total population.
According to the NHSA, as of April, most provincial-level regions in China have rolled out government-guided prices for BCI procedures. The price for a single invasive BCI implantation ranges from 6,000 to 6,600 yuan ($830 to $920), while the fee for non-invasive BCI adaptation stands at around 960 yuan ($135). This regulated pricing mechanism helps stabilize market revenue expectations for innovative medical enterprises.
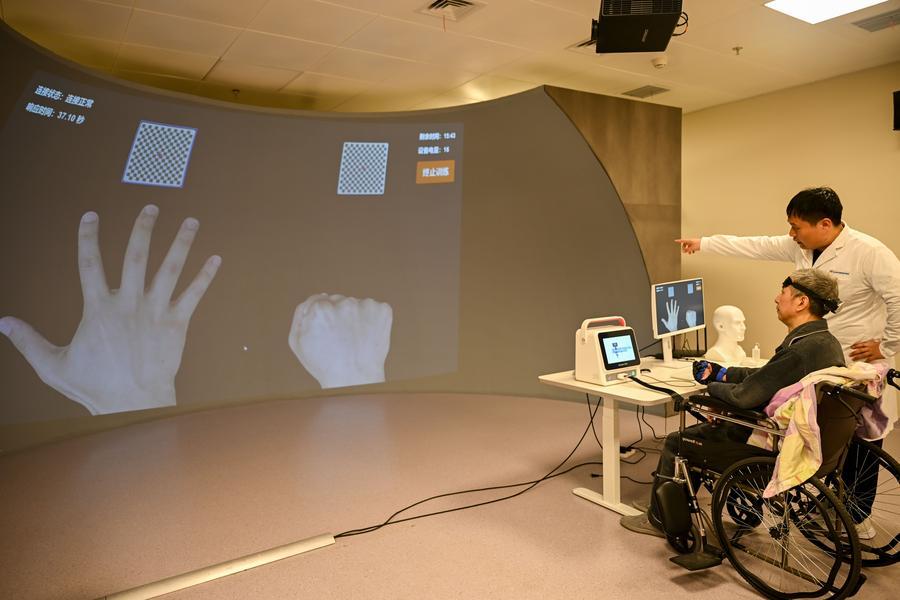
Capital flows in
Spring 2026 is also the season when global capital began to look seriously at China’s BCI industry.
Investment in the sector has surged over the first three months of the year, drawing heavy participation from major Internet companies and top-tier venture capital firms.
According to public data from IT Juzi, a China-based venture capital data platform, the BCI industry had secured 17 investment and financing deals as of mid-to-late March, totaling more than 3.8 billion yuan ($551 million). This figure far exceeds the full-year financing volume of more than 1.45 billion yuan ($212 million) recorded in 2025.
In the industrial landscape, BrainCo, based in Hangzhou, Zhejiang Province, stands out with an extensive patent portfolio and a dual-track medical plus consumer strategy. Its smart bionic hands help people with physical disabilities restore daily mobility, while home sleep monitoring devices have gained wide household popularity.
In January, newspaper Shanghai Securities News reported that BrainCo, a core enterprise among Hangzhou’s tech Six Little Dragons, submitted a confidential listing application to the Hong Kong Stock Exchange.
The company secured roughly 2 billion yuan ($292 million) in financing in early 2026, the world’s second largest funding round in the BCI sector, only behind Neuralink.
From BrainCo’s focus on rehabilitation to Chinese innovators’ continuous breakthroughs in core BCI research, the industry stays people-centric. It aims to improve quality of life for people living with neurological conditions and motor impairments, delivering new clinical and rehabilitative solutions for nervous system disorders.
On April 13, inside the operating room of Beijing Tiantan Hospital, a coin-sized internal device of Beinao No.1, China’s self-developed implantable brain-computer interface system, was precisely attached outside the patient’s dura mater via a minimally invasive cranial window.
The system was developed by NeuCyber (Beijing Xinzhida Neurotechnology Co. Ltd.), backed by state-owned ZGC Development Group. In 2022, ZGC Development Group established the Beinao Fund to support Chinese Institute for Brain Science, an institute focusing on the R&D and clinical application of brain-computer interfaces. Luo Minmin, Director of the institute, is also the founder of NeuCyber.
“This is not only a milestone showcase of homegrown BCI technologies moving toward clinical application, but also signals NeuCyber’s industry-leading capacity in system R&D, clinical adaptation and surgical standardization,” Wan Ye, an official from ZGC Capital Fund Management Co. Ltd., a subsidiary of ZGC Development Group, said in an interview with Beijing Review.
The forces shaping China’s BCI trajectory extend beyond operating rooms and into the global arena of standard-setting. In April, the ISO standard Information Technology—Brain-Computer Interfaces—Vocabulary, led by China, was officially published, becoming the world’s first unified terminology framework and consensus foundation for the BCI field.
China is making strong progress in both technological application and global standard setting. At the heart of this rapid ascent is a singular focus: turning scientific milestones into accessible healthcare tools for all—and, in the process, offering the world a replicable model for the responsible deployment of this transformative technology.
 Facebook
Facebook
 Twitter
Twitter
 Linkedin
Linkedin
 Google +
Google +










